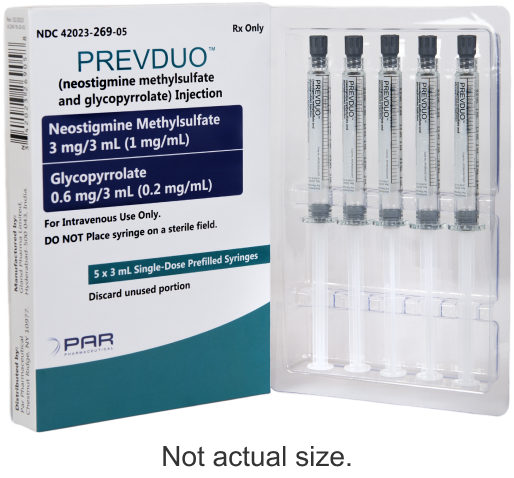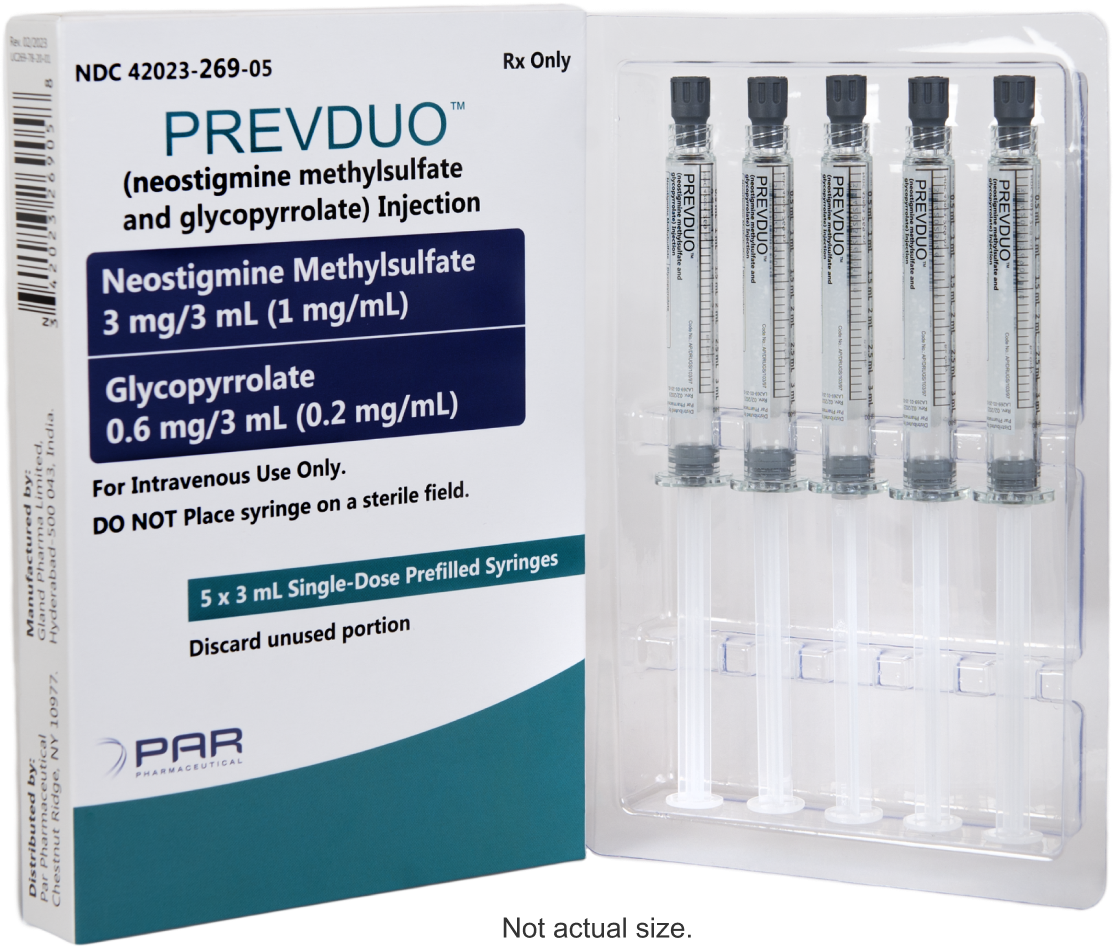
First and only FDA-approved neostigmine methylsulfate and glycopyrrolate combination product available in the US3
- Two-year shelf life4† may offer enhanced inventory management
- Does not require compounding, diluting, mixing, or transferring, which may reduce waste5
- Aligns with medication strategies of ASHP Guidelines on Preventing Medication Errors in Hospitals5
- Compatible with automated dispensing machine system
| † | When stored at the recommended storage condition of 20°C to 25°C (68°F to 77°F), protected from light. Store in carton until time of use.2 |
Recommended dosing ratio,1 simplified
When used for reversal of nondepolarizing neuromuscular blocking agents (NMBAs), the recommended dose of glycopyrrolate injection is 0.2 mg for each 1.0 mg of neostigmine methylsulfate, as stated in the FDA-approved glycopyrrolate prescribing information.6 PREVDUO® delivers the recommended dosing ratio of neostigmine methylsulfate and glycopyrrolate in one prefilled syringe.
In a survey of anesthesiologists7‡:


| ‡ | Results of an online survey from January 2023 completed by US anesthesiologists who have at least 5 anesthesiology reversal cases in an average week and who use neostigmine methylsulfate and glycopyrrolate in at least 15% of those cases (N=112). |
| § | When anesthesiologists or certified registered nurse anesthetists (CRNAs) were asked what ratio is usually used to prepare neostigmine methylsulfate and glycopyrrolate in the operating room (N=112). |
| ¶ | Among those who have received standard vials for both neostigmine methylsulfate and glycopyrrolate and were asked how they usually prepare the medication for administration, 49% of respondents reported usually using 1 syringe, 21% reported sometimes using 1 syringe, and 30% reported usually using 2 individual syringes (N=91). |
PREVDUO® product dimensions


| Product Name | NDC | Neostigmine Methylsulfate Concentration | Glycopyrrolate Concentration | Pack Size | Order Entry Number | |
|---|---|---|---|---|---|---|
| PREVDUO® (neostigmine methylsulfate and glycopyrrolate) Injection | 42023-269-05 | 3 mg/3 mL (1 mg/mL) | 0.6 mg/3 mL (0.2 mg/mL) | 5 x 3 mL single-dose prefilled syringes | Cencora10280291 Cardinal5849005 McKesson2819167 M&D281584 Henry Schein1438760 McKesson Medical-Surgical1232614 | |
M&D=Morris & Dickson; NDC=National Drug Code.